
Trusted Innovation in Bladder Control
Medtronic is the global leader in neuromodulation for bladder and bowel control — the technology used in the Altaviva™ implant — and in lifesaving devices such as cardiac pacemakers. Medtronic pioneered the first neuromodulation bladder device over 25 years ago and continues to innovate, delivering options that may help improve your quality of life.3,4,5,6
Your Team of Experts

Firstname Lastname, MD
Small bio goes here. Lorem ipsum dolor sit amet, consectetur adipisicing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.

Firstname Lastname, MD
Small bio goes here. Lorem ipsum dolor sit amet, consectetur adipisicing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.
How does it work?
A single procedure may help improve your quality of life:3
In addition to risks related to surgery, complications can include pain at the implant site, lower leg pain, infection, technical or device problems, movement of the implant, undesirable change in bowel or bladder function, or uncomfortable or unintended stimulation sensations. Please see Important Safety Information for more details. Talk with your doctor about ways to minimize these risks.
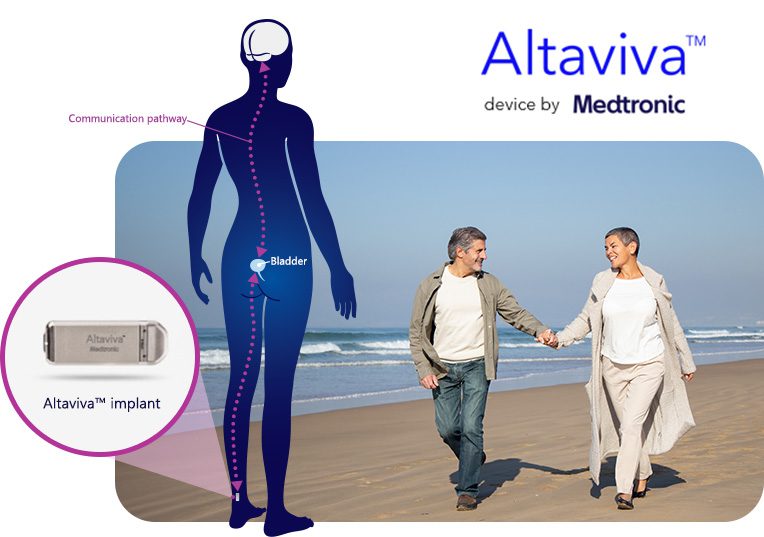
† MRI compatibility is subject to certain conditions. See approved labeling for details.
‡ Based on clinical and bench testing for expected therapy settings.
References
- Medtronic data on file: VV-TMF-841603 Statistical Report 29 Apr 2025(v1.0).
- M028929C001RevB – Clinician Therapy and Programming Guide Altaviva™ Model P7850N
- Appendix B: Clinical Study Summary. M028929C001 RevC – Clinician Therapy and Programming Guide Altaviva™ Model P7850N.
- Siegel S, Noblett K, Mangel J, et al. Five-year follow-up results of a prospective, multicenter study of patients with overactive bladder treated with sacral neuromodulation. J Urol. 2018;199(1), 229–236.
- Medtronic InterStim Therapy Clinical Summary (2018).
- Hull T, Giese C, Wexner SD, et al. Long-term durability of sacral nerve stimulation therapy for chronic fecal incontinence. Dis Colon Rectum. 2013; 56(2):234–245.
- M028929C001 RevC – Clinician Therapy and Programming Guide Altaviva™ Model P7850N.
- M028949C001 RevC – MRI Guidelines for Altaviva™ Neurostimulator
Important Safety Information
Medtronic Altaviva™ tibial neuromodulation system treats urge urinary incontinence (leakage). It should be used after you have tried other treatments such as medications and behavioral therapy, and they have not worked or you could not tolerate them.
This therapy is not for everyone. The Altaviva™ system is contraindicated (not allowed) for patients who are poor surgical candidates including patients with open wounds, sores, or damaged skin near the treatment area; current or recent history of poor blood circulation in the legs or open sores on the legs from circulation problems; physical changes or previous surgeries where the Altaviva™ device is placed. You must be able to operate or receive assistance in operating the system to be a candidate.
This therapy is not intended for patients who: are not good candidates for surgery or have conditions that make it hard to heal from wounds (such as uncontrolled diabetes, swelling in the lower leg, or nerve problems in the leg); have metal implanted within 5 cm of where the Altaviva™ device would be placed; have a current or unresolved blockage in the urinary tract caused by things like an enlarged prostate, cancer, or urethral narrowing; are allergic to any materials in the Altaviva™ device. The Altaviva™ system may affect or be affected by other implanted medical devices, including pacemakers and defibrillators. Talk to your doctor if you have a pacemaker or other implanted devices. You cannot have diathermy (deep heat treatment using shortwave or microwave electromagnetic energy) if you have an Altaviva™ device. Do not place the charger or ankle band on broken or unhealed skin. Safety and effectiveness have not been established for pregnancy; patients under the age of 18; patients with progressive, systemic neurologic disease; patients with history of urinary retention, or bilateral stimulation.
In addition to risks related to surgery, complications can include pain at the implant site or lower leg pain, infection, wound complications, nerve injury, movement of the implant, undesirable change in bowel or bladder function, uncomfortable or unintended stimulation sensations, unexpected shocking sensation, loss of therapeutic effect, discomfort when recharging, or technical or device problems.
This therapy is not for everyone. This treatment is prescribed by your doctor. Please talk to your doctor to decide whether this therapy is right for you. Your doctor should discuss all potential benefits and risks with you. Although many patients may benefit from the use of this treatment, results may vary.
For complete safety information about this treatment, please visit the Medtronic website at www.medtronic.com.
USA Rx Only. Rev 0925

